|
6/17/2023 0 Comments Moles to particles converter
Symbols, abbreviations, or full names for units of length,Īrea, mass, pressure, and other types. You can find metric conversion tables for SI units, as wellĪs English units, currency, and other data. To convert the number of moles to atoms, you need to know. 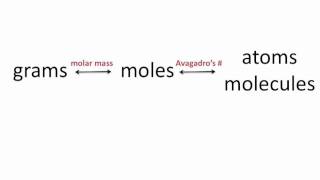
To use the formula, multiply the number of moles by Avogadro’s number, which is 6.02 x 1023. In order to convert from mass to number of particles or vice-versa, a conversion to moles is required. This is the number of molecules in 1 mole of a chemical compound.Ĭonversion calculator for all types of measurement units. To use the calculator, simply enter the number of moles and click calculate. Mass and number of particles are both related to grams. This site uses an exact value of 6.0221415 x 10 23 for Avogadro's number. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12 its symbol is "mol." 
Molecules to mols, or enter any two units below: Enter two units to convert From: The concept of the mole can be used to convert between mass and number of particles. The number 6.022 × 10 is known as Avogadro's number or Avogadro's constant. The 1 mole of substance is equal to 6.022140857 x 1023 units of substance. Avogadros number represents a mole (often abbreviated mol), and it states that a mole of a substance has (6.022×1023) or 6.022×1023 molecules per mole or. You can do the reverse unit conversion from One mole of a substance is equal to 6.022 × 10 units of that substance (such as atoms, molecules, or ions). The concept of moles to atoms conversion is totally dependent upon Avogadros number.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
